SARS-CoV-2 と COVID-19 に関するメモ・備忘録
SARS-CoV-2のNSP6タンパク質は、脳、心臓、肺の細胞経路に影響を与え、がんリスクや心血管損傷を増大させる可能性があります。NSP6が関与する主要な遺伝子「Galectin-3(LGALS3)」が10のがんの特徴すべてを促進することが明らかに。https://t.co/Wjn9kkMhzu
— Angama (@Angama_Market) January 2, 2025
この遺伝子は、がん細胞の成長、血管形成(新たな血管の形成)、炎症促進など、がんを進行させる10の特徴全てをサポートする可能性があります。https://t.co/Wjn9kkMhzu
— Angama (@Angama_Market) January 2, 2025
◆Long COVID and recovery from Long COVID: quality of life impairments and subjective cognitive decline at a median of 2 years after initial infection【bioRxiv 2024年12月30日】
Abstract
The influence of SARS-CoV-2 non-structural protein in the host’s tissue-specific complexities remains a mystery and needs more in-depth attention because of COVID-19 recurrence and long COVID. Here we investigated the influence of SARS-CoV-2 transmembrane protein NSP6 (Non-structural protein 6) in three major organs – the brain, heart, and lung in silico. To elucidate the interplay between NSP6 and host proteins, we analyzed the protein-protein interaction network of proteins regulated after SARS-CoV-2 infection and that are interacting with NSP6 interacting proteins. Pathway enrichment analyses provided global insights into biological pathways governed by differentially regulated genes in the three tissues after COVID-19 infection. Many drugs targeting hub genes of tissue-specific protein interactome were found that could be candidates for COVID-19 management. MiRNA-gene network for the tissue-specific regulated proteins was also deduced and comparing gene list targeted by SARS-CoV-2 regulated miRNAs, we found three and two common genes in the brain and lung respectively. Among the five common proteins revealed as potential therapeutic targets across the three tissues, Galectin3 (LGALS3) that was upregulated in the heart and brain after COVID-19 infection is reported to be influencing all the ten hallmarks of cancer positively and is found in multiple cancers. COVID-19 infection also causes myocardial inflammation and heart failure (HF). HF is observed to be increasing cancer incidence. Our bioinformatics and systems study hints probable effect of COVID-19 infection in cancer incidence and warrants in-depth studies in this direction and cancer surveillance especially with the present scenario of long COVID-19 and recurrent COVID-19 infections.
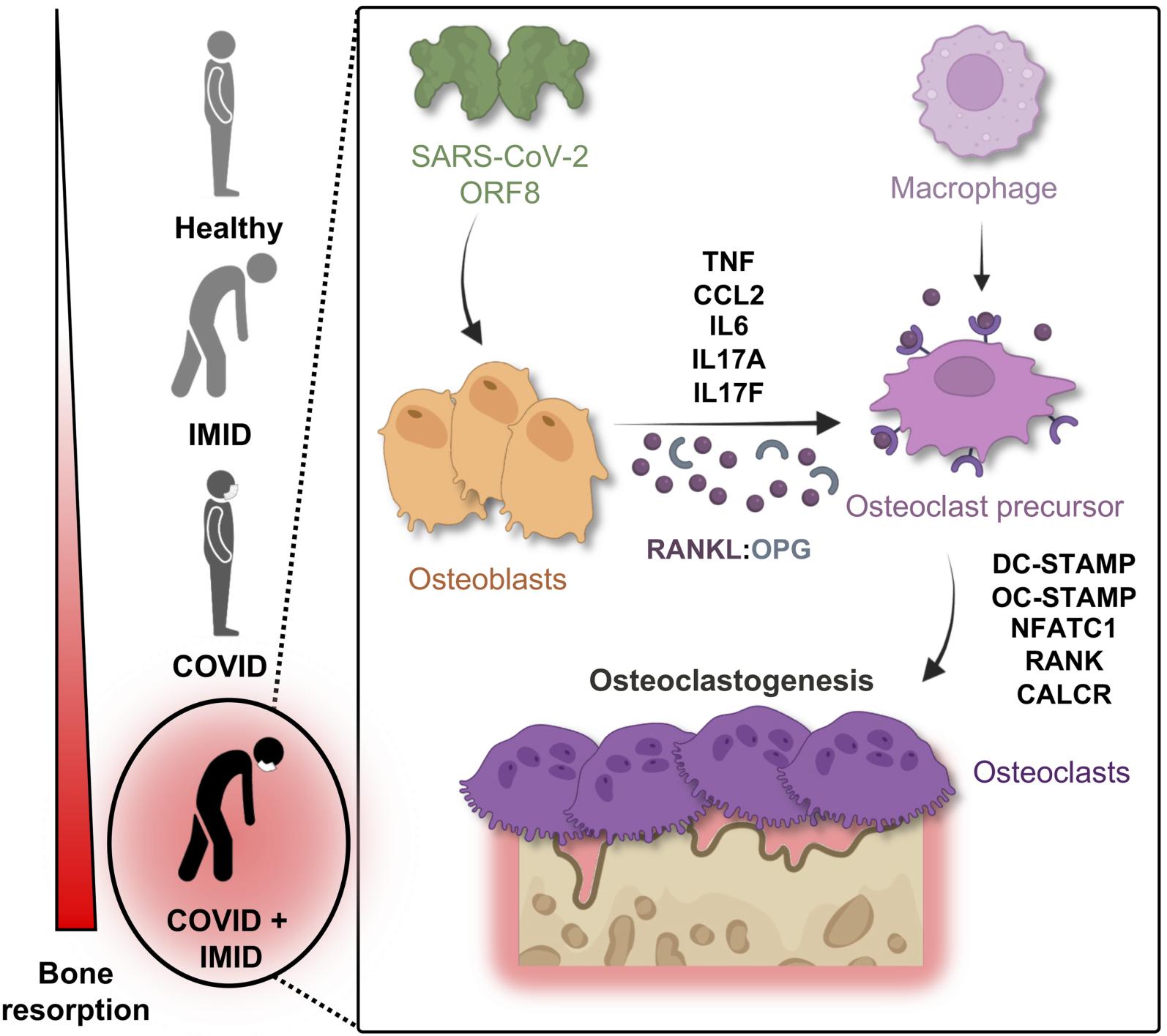
COVID-19で骨密度低下のリスクが明らかに
新研究で、SARS-CoV-2のORF8タンパク質が炎症や破骨細胞の活性化を促進し、特に関節リウマチなど免疫疾患を持つ患者で骨の破壊を引き起こすことが判明。骨を守るための新たな治療ターゲットに。— Angama (@Angama_Market) January 2, 2025
特に、関節リウマチなどの免疫疾患を持つ患者では、炎症や骨破壊がさらに進行する可能性があります。https://t.co/ktzmoyW7pa
— Angama (@Angama_Market) January 2, 2025
◆SARS-CoV-2 ORF8 drives osteoclastogenesis in preexisting immune-mediated inflammatory diseases【JCI insight 2024年12月20日】
Abstract
Patients with immune-mediated inflammatory diseases (IMIDs) like rheumatoid arthritis (RA) are at higher risk for severe COVID-19 and long-term complications in bone health. Emerging clinical evidence demonstrated that SARS-CoV-2 infection reduces bone turnover and promotes bone loss, but the mechanism underlying worsened bone health remains elusive. This study sought to identify specific immune mediators that exacerbated preexisting IMIDs after SARS-CoV-2 exposure. Plasma samples from 4 groups were analyzed: healthy, IMID only, COVID-19 only, and COVID-19 + IMID. Using high-throughput multiplexed proteomics, we profiled 1,500 protein biomarkers and identified 148 unique biomarkers in COVID-19 patients with IMIDs, including elevated inflammatory cytokines (e.g., IL-17F) and bone resorption markers. Long-term circulating SARS-CoV-2 ORF8, a virulence factor for COVID-19, was detected in the COVID + IMID group. RA was one of the most common IMIDs in our study. ORF8 treatment of RA-derived human osteoblasts (RA-hOBs) increased levels of inflammatory (TNF, IL6, CCL2) and bone resorption (RANKL/osteoprotegerin ratio) markers compared with healthy controls. Supernatants from ORF8-treated RA-hOBs drove the differentiation of macrophages into osteoclast-like cells. These findings suggest that SARS-CoV-2 exposure can exacerbate IMIDs through ORF8-driven inflammation and osteoclastogenesis, highlighting potential therapeutic targets for managing COVID-19–induced bone pathologies.
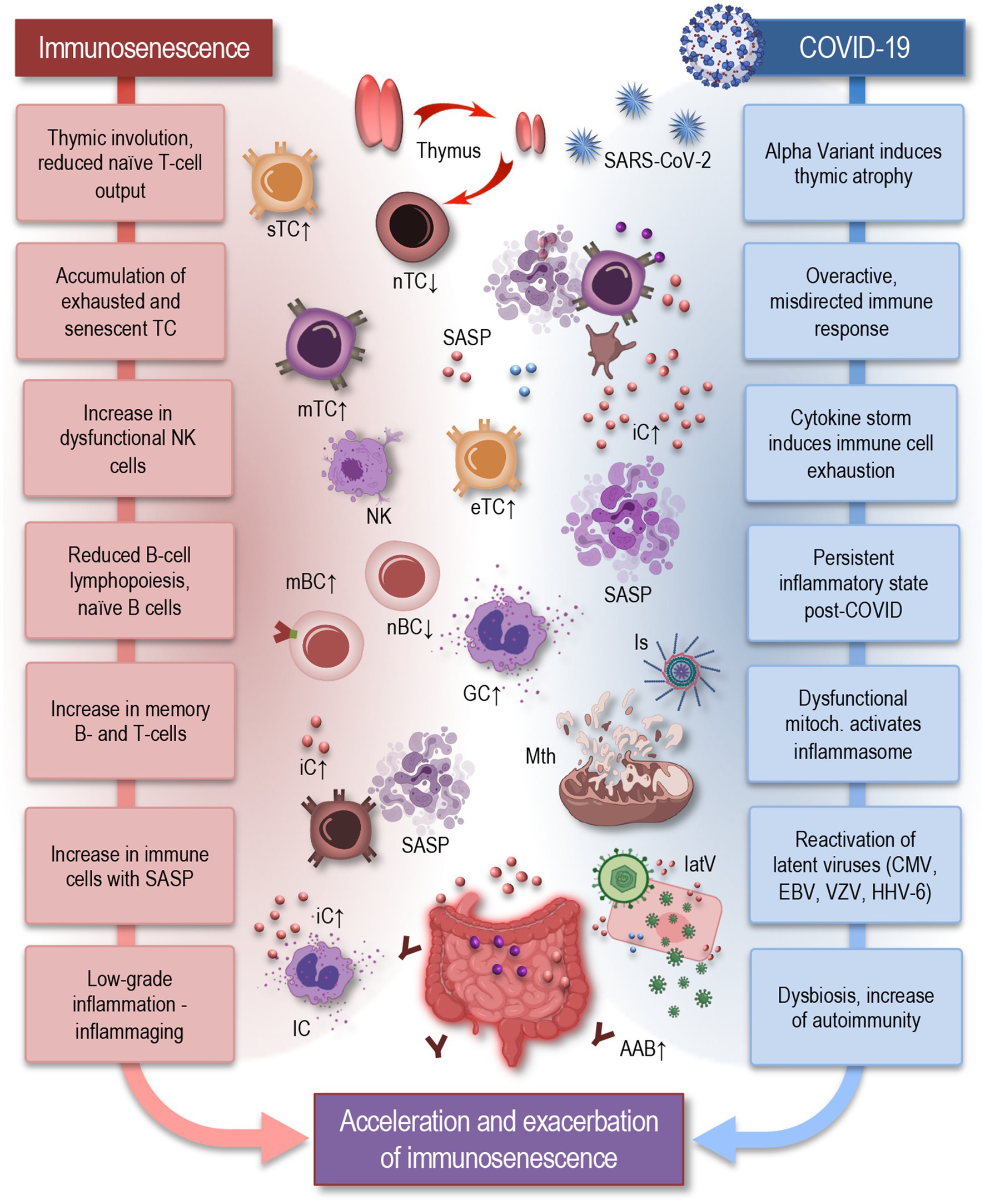
COVID-19が脳の加齢を加速する可能性
SARS-CoV-2は慢性炎症を引き起こし、免疫老化や脳炎症を促進。長期的な神経認知障害や神経変性疾患のリスクが上昇。特に脳の加齢が進行する可能性が示唆されています。科学者はこれを「加速された神経免疫老化」と表現。https://t.co/LSiCSR6ImL— Angama (@Angama_Market) January 2, 2025
長期的なリスク:脳霧、記憶力低下、注意力欠如が特に長引くケースで報告されています。
重要なメカニズム:慢性的な血液脳関門の障害、炎症シグナルの持続、そして脳内での神経炎症が影響しています。COVID-19感染者が早期老化のリスクにさらされる可能性を示唆。https://t.co/LSiCSR6ImL
— Angama (@Angama_Market) January 2, 2025
◆The impact of COVID-19 on accelerating of immunosenescence and brain aging【Frontiers in Cellular Neuroscience 2024年12月10日】
The COVID-19 pandemic, caused by the novel coronavirus SARS-CoV-2, has profoundly impacted global health, affecting not only the immediate morbidity and mortality rates but also long-term health outcomes across various populations. Although the acute effects of COVID-19 on the respiratory system have initially been the primary focus, it is increasingly evident that the virus can have significant impacts on multiple physiological systems, including the nervous and immune systems. The pandemic has highlighted the complex interplay between viral infection, immune aging, and brain health, that can potentially accelerate neuroimmune aging and contribute to the persistence of long COVID conditions. By inducing chronic inflammation, immunosenescence, and neuroinflammation, COVID-19 may exacerbate the processes of neuroimmune aging, leading to increased risks of cognitive decline, neurodegenerative diseases, and impaired immune function. Key factors include chronic immune dysregulation, oxidative stress, neuroinflammation, and the disruption of cellular processes. These overlapping mechanisms between aging and COVID-19 illustrate how the virus can induce and accelerate aging-related processes, leading to an increased risk of neurodegenerative diseases and other age-related conditions. This mini-review examines key features and possible mechanisms of COVID-19-induced neuroimmune aging that may contribute to the persistence and severity of long COVID. Understanding these interactions is crucial for developing effective interventions. Anti-inflammatory therapies, neuroprotective agents, immunomodulatory treatments, and lifestyle interventions all hold potential for mitigating the long-term effects of the virus. By addressing these challenges, we can improve health outcomes and quality of life for millions affected by the pandemic.
ポストCOVID症候群とME/CFSに関与する自己抗体を特定
新研究で、EBウイルス抗原に由来する自己抗体が、疲労、認知機能低下、自律神経障害に関連。これらの抗体が自己免疫応答を引き起こし、ミトコンドリアや血管機能に影響を与える可能性が示唆されました。 https://t.co/DFhmqMGt0y— Angama (@Angama_Market) January 3, 2025
これらの自己抗体は、他のウイルス抗原と交差反応し、ミトコンドリアや血管の機能を乱す可能性があります。https://t.co/DFhmqMGt0y
— Angama (@Angama_Market) January 3, 2025
◆Long COVID and recovery from Long COVID: quality of life impairments and subjective cognitive decline at a median of 2 years after initial infection【bioRxiv 2024年12月30日】
Abstract
Background Epstein-Barr virus (EBV) infection is a known trigger and risk factor for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and post-COVID syndrome (PCS). In previous studies, we found enhanced IgG reactivity to EBV EBNA4 and EBNA6 arginine-rich sequences in postinfectious ME/CFS (piME/CFS).
Objective This study aims to investigate IgG responses to arginine-rich (poly-R) EBNA4 and EBNA6 sequences and homologous human sequences in PCS and ME/CFS.
Methods The IgG responses against poly-R EBNA4 and EBNA6 and corresponding homologous human 15-mer peptides and respective full-length proteins were analyzed using a cytometric bead array (CBA) and a multiplex dot-blot assay. Sera of 45 PCS patients diagnosed according to WHO criteria, with 26 patients fulfilling the Canadian Consensus criteria for ME/CFS (pcME/CFS), 36 patients with non-COVID post-infectious ME/CFS (piME/CFS), and 34 healthy controls (HC) were investigated.
Results Autoantibodies to poly-R peptide sequences of the neuronal antigen SRRM3, the ion channel SLC24A3, TGF-β signaling regulator TSPLY2, angiogenic regulator TSPYL5, as well as to full-length α-adrenergic receptor (ADRA) proteins were more frequent in patients. Several autoantibodies were positively associated with key symptoms of autonomic dysfunction, fatigue, cognition, and pain.
スパイクたんぱく質は、感染後も頭蓋骨・髄膜・脳の軸に残り、神経炎症や神経変性を引き起こす可能性が示されました。ACE2経路を介した炎症の活性化が影響を悪化させ、脳卒中や外傷性脳損傷のリスクを高める可能性も。ワクチン接種は負荷を軽減しますが、完全な排除には至らずhttps://t.co/OXCZzSmw7s
— Angama (@Angama_Market) January 7, 2025
◆Persistence of spike protein at the skull-meninges-brain axis may contribute to the neurological sequelae of COVID-19【Cell Host & Microbe 2024年12月11日】
Summary
SARS-CoV-2 infection is associated with long-lasting neurological symptoms, although the underlying mechanisms remain unclear. Using optical clearing and imaging, we observed the accumulation of SARS-CoV-2 spike protein in the skull-meninges-brain axis of human COVID-19 patients, persisting long after viral clearance. Further, biomarkers of neurodegeneration were elevated in the cerebrospinal fluid from long COVID patients, and proteomic analysis of human skull, meninges, and brain samples revealed dysregulated inflammatory pathways and neurodegeneration-associated changes. Similar distribution patterns of the spike protein were observed in SARS-CoV-2-infected mice. Injection of spike protein alone was sufficient to induce neuroinflammation, proteome changes in the skull-meninges-brain axis, anxiety-like behavior, and exacerbated outcomes in mouse models of stroke and traumatic brain injury. Vaccination reduced but did not eliminate spike protein accumulation after infection in mice. Our findings suggest persistent spike protein at the brain borders may contribute to lasting neurological sequelae of COVID-19.
ロングCOVID患者では、ミトコンドリアの機能異常が確認され、エネルギー代謝の破綻が疲労や「ブレインフォグ」などの症状を引き起こしています。特にccf-mtDNA(循環型細胞外ミトコンドリアDNA)の減少が重要なバイオマーカーとして注目され、治療法開発の手がかりの可能性。https://t.co/iZnLVVz7o7
— Angama (@Angama_Market) January 7, 2025
◆Novel biomarkers of mitochondrial dysfunction in Long COVID patients【Cell Host & Microbe 2024年11月4日】
Abstract
Coronavirus disease 2019 (COVID-19) can lead to severe acute respiratory syndrome, and while most individuals recover within weeks, approximately 30–40% experience persistent symptoms collectively known as Long COVID, post-COVID-19 syndrome, or post-acute sequelae of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection (PASC). These enduring symptoms, including fatigue, respiratory difficulties, body pain, short-term memory loss, concentration issues, and sleep disturbances, can persist for months. According to recent studies, SARS-CoV-2 infection causes prolonged disruptions in mitochondrial function, significantly altering cellular energy metabolism. Our research employed transmission electron microscopy to reveal distinct mitochondrial structural abnormalities in Long COVID patients, notably including significant swelling, disrupted cristae, and an overall irregular morphology, which collectively indicates severe mitochondrial distress. We noted increased levels of superoxide dismutase 1 which signals oxidative stress and elevated autophagy-related 4B cysteine peptidase levels, indicating disruptions in mitophagy. Importantly, our analysis also identified reduced levels of circulating cell-free mitochondrial DNA (ccf-mtDNA) in these patients, serving as a novel biomarker for the condition. These findings underscore the crucial role of persistent mitochondrial dysfunction in the pathogenesis of Long COVID. Further exploration of the cellular and molecular mechanisms underlying post-viral mitochondrial dysfunction is critical, particularly to understand the roles of autoimmune reactions and the reactivation of latent viruses in perpetuating these conditions. This comprehensive understanding could pave the way for targeted therapeutic interventions designed to alleviate the chronic impacts of Long COVID. By utilizing circulating ccf-mtDNA and other novel mitochondrial biomarkers, we can enhance our diagnostic capabilities and improve the management of this complex syndrome.
MERS-CoVとSARS-CoV-2の膜タンパク質でβシート構造を確認。
βシートサンドイッチ領域が両ウイルスで共通して観察され、これが「プリオン様挙動」の可能性を示唆。高濃度やストレス環境下での凝集や細胞損傷への関与が懸念され、さらなる研究が必要。https://t.co/EX6xVBklQn— Angama (@Angama_Market) January 7, 2025
◆Conformational dynamics of the membrane protein of MERS-CoV in comparison with SARS-CoV-2 in ERGIC complex【Taylor & Francis Online 2025年1月5日】
Abstract
The present study explores the conformational dynamics of the membrane protein of Middle East Respiratory Syndrome Coronavirus (MERS-CoV) within the Endoplasmic Reticulum-Golgi Intermediate Compartment (ERGIC) complex using an all-atomistic molecular dynamics simulation approach. Significant structural changes were observed in the N-terminal, C-terminal, transmembrane, and beta-sheet sandwich domains of the MERS-CoV membrane protein. This study also highlights the structural similarities between the MERS-CoV and the SARS-CoV-2 membrane proteins, particularly in how both exhibit a distinct kink in the transmembrane helix caused by aromatic residue-lipid interactions. A structural expansion below the transmembrane and above the beta-sheet sandwich domain within the dimer was observed in all the M-proteins. This site on the beta-sheet sandwich domains near the C-terminal end could serve as a potential drug-binding site. Notably, a stable helical structure was identified in the C-terminal domain of the MERS-CoV membrane protein, whereas a proper secondary structural conformation was not observed in the SARS-CoV-2 membrane protein. Further, the SARS-CoV-2 membrane protein exhibited stronger binding to the lipid bilayer than the MERS-CoV, indicating its greater structural stability within the ERGIC complex. The structural similarity between the membrane protein of MERS-CoV and SARS-CoV-2 suggests the feasibility of employing a common inhibitor against these beta-coronaviruses. Furthermore, this analysis enhances our understanding of the membrane protein’s interactions with proteins and lipids, paving the way for therapeutic developments against these viruses.
乳幼児期(生後3年)の感染症エピソードが多い子どもは、成長後(10〜13歳)に中等度〜重度の感染症や抗生物質の使用リスクが増加することが判明。特に風邪、肺炎、ウイルス感染が影響。都市部では感染リスクが高い。若年時の感染管理が健康寿命に直結する可能性。https://t.co/caVW6bdVef
— Angama (@Angama_Market) January 8, 2025
◆Burden of Infections in Early Life and Risk of Infections and Systemic Antibiotics Use in Childhood【JAMA Network 2025年1月6日】
Key Points
Question Does early-life infection burden track throughout childhood and into adolescence?
Findings In this cohort study of 614 children with daily diary information on common infection load in early life, the overall infection burden in early life was associated with increased risks of moderate to severe infections and antibiotic treatments later in childhood.
Meaning These findings are important for prognosis and follow-up of children experiencing a high burden of common infections in early life.
Abstract
Importance A high infection burden in early childhood is common and a risk factor for later disease development. However, longitudinal birth cohort studies investigating early-life infection burden and later risk of infection and antibiotic episodes are lacking.
Objective To investigate whether early-life infection burden is associated with a later risk of infection and systemic antibiotic treatment episodes in childhood.
Design, Setting, and Participants This longitudinal cohort study of children from birth to age 10 or 13 years included data from the Danish population-based Copenhagen Prospective Studies on Asthma in Childhood (COPSAC) birth cohort between November 2008 to November 2010. Children were monitored for infection diagnoses and systemic antibiotic prescriptions from national databases until February 1, 2024, by which time they had completed the age 10- or 13-year visit. Children with immune deficiencies or congenital diseases were excluded.
Exposures Daily diary-registered common infection episodes of cold, acute otitis media, tonsillitis, pneumonia, gastroenteritis, and fever episodes from birth to 3 years.
Main Outcome and Measures After age 3 years, the incidence of moderate to severe infection diagnoses and systemic antibiotic prescriptions were estimated using adjusted incidence rate ratios (AIRRs) calculated from quasi-Poisson regression models. All analyses were adjusted for social and environmental confounders.
Results A total of 614 children (317 male [51.6%]) with diary data from birth to 3 years had completed follow-up until age 10 or 13 years. No differences in baseline characteristics between the children having vs not having available diary data were noted. Children with a high vs low burden of diary-registered infections between birth and 3 years (ie, equal to and above vs below the median of 16) had an increased risk of later moderate to severe infections (181 vs 87 episodes; AIRR, 2.39; 95% CI, 1.52-3.89) and systemic antibiotic treatments (799 vs 623 episodes; AIRR, 1.34; 95% CI, 1.07-1.68) until age 10 or 13 years. Each diary infection episode also increased the later risk of moderate to severe infections (AIRR, 1.05; 95% CI, 1.02-1.08) and systemic antibiotic treatments (AIRR, 1.02; 95% CI, 1.01-1.04). Subtype analyses showed significant associations between each cold, acute otitis media, pneumonia, gastroenteritis, and fever episode between birth and 3 years and risk of later moderate to severe infections or systemic antibiotic treatments.
Conclusions and relevance This longitudinal cohort study suggests that early-life infection burden may continue throughout childhood and is associated with later antibiotic treatments independent of social and environmental risk factors. These findings are important for prognosis and follow-up of children experiencing a high burden of common infections in early life.
無症状や軽症の若年層(18〜25歳)でも、コロナウイルス感染後3〜12ヶ月で肺機能、下肢筋力、身体機能の低下が確認され、特に6ヶ月以降進行的に悪化。観察期間は12ヶ月までで、完全回復は確認されず。長期的な経過観察と予防が必要。https://t.co/5PebOW7AP6 #mdpimedicina
— Angama (@Angama_Market) January 8, 2025
◆Exploring Medium- and Long-Term Respiratory and Functional Sequelae in Young Adults Post-COVID-19【MDPI 2025年1月7日】
Abstract
Background and Objectives: Long COVID-19 syndrome may cause difficulties in functionality during daily life in young people. Our objective was to investigate the respiratory and functional sequelae in young adults with asymptomatic or mild COVID-19 compared with healthy peers 3–6 months and 6–12 months after COVID-19 infection. Materials and Methods: Participants aged 18–25 who had COVID-19 within the last 3–6 months (Post-COVID Group 1, n = 25) and 6–12 months (Post-COVID Group 2, n = 25) and age–gender-matched healthy controls (n = 25) were included in this study. Respiratory functions and muscle strength were measured. Physical function was assessed with 6 min walking test (6MWT) and an Incremental Shuttle Walk Test (ISWT). The 1 min sit-to-stand test (1-MSTST) and hand grip strength (HGS) were used to assess muscle performance. Fatigue and dyspnea severity were questioned. Results: The FVC%pred (p = 0.023) and MEP (p = 0.034) were higher, and 1-MSTST repetitions were lower in Post-COVID Group-1 compared to Post-COVID Group-2 (p = 0.029). The PEF%pred (p = 0.025), MEP (p = 0.001), and ISWT distance were lower in Post-COVID Group-2 compared to healthy controls. The number of 1-MSTST repetitions and 6MWT distance were lower in Post-COVID Group-1 (p = 0.003, p = 0.001) and Post-COVID Group-2 (p = 0.003, p = 0.017) than in healthy controls. Exercise-induced blood lactate change during the ISWT, HGS, fatigue, and dyspnea were not significantly different between post-COVID groups and healthy controls. Conclusions: Young adults who pass asymptomatic or mild SARS-CoV-2 infection exhibit a decline in FVC%pred, PEF%pred, lower extremity muscle performance, and physical function within 3–6 months. In addition, the deterioration in respiratory and physical functions becomes apparent within 6–12 months.
ロングコロナの蔓延や加速する気候変動などを前にするとストレートに考えることができなくなるような状態をよく「サーキットブレーカーが落ちる」と表現することがありますが、これは実際に海馬のCA3というサブ部位に興奮性の神経伝達物質受容体が密集していて、活発化しすぎると”焼けてしまう”
— Angama (@Angama_Market) January 9, 2025
回避行動を優先するために扁桃体とは”優先回線”で繋がっていて、CA3が不調になると恐怖感が増幅されます。また、断片から全体を構築する働きが過剰になり、恐怖の想像がコントロールを失うPTSDや統合失調症とも関連しています。興奮性が高い部位であること、他部位との接続が多いこと、
— Angama (@Angama_Market) January 9, 2025
CA3に興奮性受容体が多目に密集している、あるいは抑制性受容体が少ない、あるいはミトコンドリアが何らかの理由で少ない場合、ショックに顕著に脆弱になります。
— Angama (@Angama_Market) January 9, 2025
コロナウイルスの話題や異常気象の議論になるとコントロールが効かなくなるためにこういった問題が公で話しづらくなる背景には、こういった脳神経メカニズムがあると思います。
— Angama (@Angama_Market) January 9, 2025
人間の体は、長い飢えや氷河期などの自然からの抑圧に対抗するため、「発散を増幅する」方向で進化しています。興奮性受容体が抑制性よりも遥かに多いことや、免疫が暴走すると制御性を圧倒してしまう自己免疫疾患などで顕著です。
— Angama (@Angama_Market) January 9, 2025
自然からの圧力があることでバランスが取れるシステムなので、自力でバランスを取り戻すには医学や科学の助けが必要です。
— Angama (@Angama_Market) January 9, 2025
「感染繰り返し」がコロナ後遺症リスクを増大
NY研究:再感染者の30.8%が後遺症発症、単回感染の16.7%より高リスク。再感染は1.41倍のリスク増加。
複数回感染が体内ウイルスの蓄積により症状を慢性化させる可能性。ME/CFSと類似のメカニズムも示唆。
感染回避と予防策が鍵。https://t.co/QloCI0gznI— Angama (@Angama_Market) January 9, 2025
◆SARS-COV-2 re-infection and incidence of post-acute sequelae of COVID-19 (PASC) among essential workers in New York: a retrospective cohort study【THE LANCET Regional Health Americas 2025年1月7日】
Summary
Background
After surviving Coronavirus Disease 2019 (COVID-19), some people develop symptoms known as post-acute sequelae of COVID-19 (PASC). PASC is an emerging phenomenon yet to be fully understood, and identifying risk factors has been challenging. This study investigated the association between the number of COVID-19 episodes and the incidence of PASC among essential workers.
Methods
We analyzed data from 2511 essential workers, mainly first responders, with confirmed polymerase chain reaction, antibody, or antigen-positive test results for SARS-CoV-2 infection from March 2020 to February 2024. Data were collected through in-person questionnaires and surveys sent via text and email, internal medical records, follow-up calls, and external medical records. Participants who reported continuation or the development of new symptoms three months after the initial SARS-CoV-2 infection, with symptoms lasting for at least two months, were categorized as having PASC, while those without any COVID-19 or whose symptoms resolved were classified as non-PASC. PASC was common in this cohort so we used a Poisson regression model to compute multivariable-adjusted Relative Risk (RR) for the association between risk of PASC and SARS-CoV-2 re-infection, severity, and vaccination status at first infection.
Findings
A total of 475 (prevalence = 18.9%, [95% confidence interval] = [17.4–20.5]) PASC patients were identified. The mean (standard deviation (SD)) age of participants who experienced PASC (54.8 (7.2) years) was similar to those who did not (54.2 (7.4) years). There were 403 (16.1% [14.6–17.5]) participants who experienced multiple instances of COVID-19. After adjusting for relevant demographic, lifestyle, and clinical variables, we found a significant association between the risk of experiencing PASC and multiple SARS-COV-2 infections (RR = 1.41 [1.14–1.74]), severe COVID-19 (RR = 3.17 [2.41–4.16]), and being unvaccinated at first infection (RR = 3.29 [2.46–4.41]).
Interpretation
Although the pathogenetic mechanism for PASC remains unclear, identifying risk factors such as lack of vaccination or re-infection can assist in better understanding and managing the condition.
新型コロナウイルスは人間の肝細胞を直接感染させ、細胞死誘発
ACE2とTMPRSS2を介して感染し、肝臓内でウイルスが複製・拡散。デルタやオミクロンは、早いが軽度の細胞死を引き起こす一方で、免疫反応が肝臓損傷を悪化させる可能性も。肝疾患患者にとって新たな課題— Angama (@Angama_Market) January 9, 2025
◆SARS-CoV-2 Productively Infects Human Hepatocytes and Induces Cell Death【WILEY Online Library 2025年1月6日】
ABSTRACT
SARS-CoV-2 infection is accompanied by elevated liver enzymes, and patients with pre-existing liver conditions experience more severe disease. While it was known that SARS-CoV-2 infects human hepatocytes, our study determines the mechanism of infection, demonstrates viral replication and spread, and highlights direct hepatocyte damage. Viral replication was readily detectable upon infection of primary human hepatocytes and hepatoma cells with the ancestral SARS-CoV-2, Delta, and Omicron variants. Hepatocytes express the SARS-CoV-2 receptor ACE2 and the host cell protease TMPRSS2, and knocking down ACE2 and TMPRSS2 impaired SARS-CoV-2 infection. Progeny viruses released from infected hepatocytes showed the typical coronavirus morphology by electron microscopy and proved infectious when transferred to fresh cells, indicating that hepatocytes can contribute to virus spread. Importantly, SARS-CoV-2 infection rapidly induced hepatocyte death in a replication-dependent fashion, with the Omicron variant showing faster onset but less extensive cell death. C57BL/6 wild-type mice infected with a mouse-adapted SARS-CoV-2 strain showed high levels of viral RNA in liver and lung tissues. ALT peaked when viral RNA was cleared from the liver. Liver histology revealed profound tissue damage and immune cell infiltration, indicating that direct cytopathic effects of SARS-CoV-2 and immune-mediated killing of infected hepatocytes contribute to liver pathology.
コロナ感染が子ども・若者の精神健康にリスク
米研究:陽性児童(5-12歳)で不安、ADHD、OCD、自閉症が増加。若者(12-20歳)では不安、うつ、自殺傾向が上昇。感染後28-179日に観察。
コロナは単なるストレスでなく直接リスク要因の可能性。早期治療と予防が重要。
https://t.co/r87EiBR02B— Angama (@Angama_Market) January 9, 2025
◆Does SARS-CoV-2 Infection Increase Risk of Neuropsychiatric and Related Conditions? Findings from Difference-in-Differences Analyses【Research Square 2025年1月7日】
Abstract
The COVID-19 pandemic has been associated with increased neuropsychiatric conditions in children and youths, with evidence suggesting that SARS-CoV-2 infection may contribute additional risks beyond pandemic stressors. This study aimed to assess the full spectrum of neuropsychiatric conditions in COVID-19 positive children (ages 5–12) and youths (ages 12–20) compared to a matched COVID-19 negative cohort, accounting for factors influencing infection risk. Using EHR data from 25 institutions in the RECOVER program, we conducted a retrospective analysis of 326,074 COVID-19 positive and 887,314 negative participants matched for risk factors and stratified by age. Neuropsychiatric outcomes were examined 28 to 179 days post-infection or negative test between March 2020 and December 2022. SARS-CoV-2 positivity was confirmed via PCR, serology, or antigen tests, while negativity required negative test results and no related diagnoses. Risk differences revealed higher frequencies of neuropsychiatric conditions in the COVID-19 positive cohort. Children faced increased risks for anxiety, OCD, ADHD, autism, and other conditions, while youths exhibited elevated risks for anxiety, suicidality, depression, and related symptoms. These findings highlight SARS-CoV-2 infection as a potential contributor to neuropsychiatric risks, emphasizing the importance of research into tailored treatments and preventive strategies for affected individuals.
コロナ後遺症患者の生活の質に影響する要因
米研究:後遺症患者53人(76%女性、平均年齢54歳)の49%で認知障害を確認。生活の質(QoL)は疲労(7%)と心理的苦痛(11%)が主要要因。感染から8.7か月後評価。疲労と苦痛を治療対象とすべき可能性。さらなる研究が必要。 https://t.co/8zbNyS0gSV— Angama (@Angama_Market) January 10, 2025
やはり脳の問題は脳自身では気づきにくく、自覚のない認知障害が水面下で広がっているのではないかと思います。
— Angama (@Angama_Market) January 10, 2025
◆Factors associated with quality of life in long-COVID syndrome【International Journal of Rehabilitation Research 2024年12月25日】
Abstract
Approximately 10% of patients experience persistent symptoms following COVID-19, known as long-COVID syndrome. This cross-sectional study explored factors of quality of life (QoL) in 53 long-COVID patients. QoL was measured using the World Health Organization-Five Well-Being Index, fatigue with the Fatigue Visual Analogue Scale, and psychological health with the Depression-Anxiety-Stress-21 questionnaire. Six neuropsychological tests assessed information processing speed, verbal memory, visual memory, working memory, attention, language, fluency, recall, and visuospatial function with a composite score calculated by averaging z scores. Patients (76% female, mean age: 54.1 years) were assessed 8.7 months postinfection. Cognitive impairment, present in 49% of the sample, was not associated with QoL. In multiple linear regression, gender, fatigue, and psychological distress accounted for 42% of QoL variance, with fatigue and distress contributing 7% and 11%, respectively. Further studies are needed to determine if fatigue and psychological distress are causally related to QoL in long-COVID and could be treatment targets.
COVID-19後の成人でME/CFS(筋痛性脳脊髄炎/慢性疲労症候群)発症率が約5倍に増加。感染から6か月以上後の追跡調査で、11,785人中4.5%がME/CFS診断基準を満たす一方、非感染者では0.6%。疲労や倦怠感の症状が日常生活に与える影響を軽視できず。感染予防が極めて重要。https://t.co/LkG5EXEn3D
— Angama (@Angama_Market) January 14, 2025
◆Incidence and Prevalence of Post-COVID-19 Myalgic Encephalomyelitis: A Report from the Observational RECOVER-Adult Study【SPRINGER NATURE 2025年1月13日】
Abstract
Background
Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) may occur after infection. How often people develop ME/CFS after SARS-CoV-2 infection is unknown.
Objective
To determine the incidence and prevalence of post-COVID-19 ME/CFS among adults enrolled in the Researching COVID to Enhance Recovery (RECOVER-Adult) study.
Design, Setting, and Participants
RECOVER-Adult is a longitudinal observational cohort study conducted across the U.S. We included participants who had a study visit at least 6 months after infection and had no pre-existing ME/CFS, grouped as (1) acute infected, enrolled within 30 days of infection or enrolled as uninfected who became infected (n=4515); (2) post-acute infected, enrolled greater than 30 days after infection (n=7270); and (3) uninfected (1439).
Measurements
Incidence rate and prevalence of post-COVID-19 ME/CFS based on the 2015 Institute of Medicine ME/CFS clinical diagnostic criteria.
Results
The incidence rate of ME/CFS in participants followed from time of SARS-CoV-2 infection was 2.66 (95% CI 2.63–2.70) per 100 person-years while the rate in matched uninfected participants was 0.93 (95% CI 0.91–10.95) per 100 person-years: a hazard ratio of 4.93 (95% CI 3.62–6.71). The proportion of all RECOVER-Adult participants that met criteria for ME/CFS following SARS-CoV-2 infection was 4.5% (531 of 11,785) compared to 0.6% (9 of 1439) in uninfected participants. Post-exertional malaise was the most common ME/CFS symptom in infected participants (24.0%, 2830 of 11,785). Most participants with post-COVID-19 ME/CFS also met RECOVER criteria for long COVID (88.7%, 471 of 531).
Limitations
The ME/CFS clinical diagnostic criteria uses self-reported symptoms. Symptoms can wax and wane.
Conclusion
ME/CFS is a diagnosable sequela that develops at an increased rate following SARS-CoV-2 infection. RECOVER provides an unprecedented opportunity to study post-COVID-19 ME/CFS.
新型コロナウイルス(SARS-CoV-2)のウイルス粒子が、急性期終了後も体内で長期間残存することが判明。
血漿・唾液中で最大68日間、糞便中では3ヶ月以上検出。感染後もウイルスの持続的な存在が、後遺症や長期的な健康リスクに関与する可能性。 #LongCOVID— Angama (@Angama_Market) January 14, 2025
◆Ultrasensitive detection of intact SARS-CoV-2 particles in complex biofluids using microfluidic affinity capture【Science Advances 2025年1月10日】
Abstract
Measuring virus in biofluids is complicated by confounding biomolecules coisolated with viral nucleic acids. To address this, we developed an affinity-based microfluidic device for specific capture of intact severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Our approach used an engineered angiotensin-converting enzyme 2 to capture intact virus from plasma and other complex biofluids. Our device leverages a staggered herringbone pattern, nanoparticle surface coating, and processing conditions to achieve detection of as few as 3 viral copies per milliliter. We further validated our microfluidic assay on 103 plasma, 36 saliva, and 29 stool samples collected from unique patients with COVID-19, showing SARS-CoV-2 detection in 72% of plasma samples. Longitudinal monitoring in the plasma revealed our device’s capacity for ultrasensitive detection of active viral infections over time. Our technology can be adapted to target other viruses using relevant cell entry molecules for affinity capture. This versatility underscores the potential for widespread application in viral load monitoring and disease management.
重症COVID-19回復者の免疫細胞におけるミトコンドリア機能低下が明らかに。代謝と免疫が長期にわたり異常を示し、特定のサイトカイン(IL-1β増加、IL-27減少)が関連。軽症回復者との差異は、ミトコンドリアの融合/分裂異常に加え、エネルギー代謝の低下も。https://t.co/cWZ8cCwiPr
— Angama (@Angama_Market) January 14, 2025
◆Long-term mitochondrial and metabolic impairment in lymphocytes of subjects who recovered after severe COVID-19【SPRINGER NATURE 2025年1月10日】
Abstract
The underlying mechanisms explaining the differential course of SARS-CoV-2 infection and the potential clinical consequences after COVID-19 resolution have not been fully elucidated. As a dysregulated mitochondrial activity could impair the immune response, we explored long-lasting changes in mitochondrial functionality, circulating cytokine levels, and metabolomic profiles of infected individuals after symptoms resolution, to evaluate whether a complete recovery could be achieved. Results of this pilot study evidenced that different parameters of aerobic respiration in lymphocytes of individuals recuperated from a severe course lagged behind those shown upon mild COVID-19 recovery, in basal conditions and after simulated reinfection, and they also showed altered glycolytic capacity. The severe groups showed trends to enhanced superoxide production in parallel to lower OPA1-S levels. Unbalance of pivotal mitochondrial fusion (MFN2, OPA1) and fission (DRP1, FIS1) proteins was detected, suggesting a disruption in mitochondrial dynamics, as well as a lack of structural integrity in the electron transport chain. In serum, altered cytokine levels of IL-1β, IFN-α2, and IL-27 persisted long after clinical recovery, and growing amounts of the latter after severe infection correlated with lower basal and maximal respiration, ATP production, and glycolytic capacity. Finally, a trend for higher circulating levels of 3-hydroxybutyrate was found in individuals recovered after severe compared to mild course. In summary, long after acute infection, mitochondrial and metabolic changes seem to differ in a situation of full recovery after mild infection versus the one evolving from severe infection.